About Us

Our group is based in Turku PET Centre, University of Turku and Turku University Hospital. Our research focuses on developing radiopharmaceuticals for the PET imaging of various inflammatory diseases. (Roivainen and Saraste Group members with principle investigators : Prof. Anne Roivainen (middle left) and Prof. Antti Saraste (next to Anne).
Research:
Timely identification and localization of inflammatory foci are critical for adequate treatment of patients and current methodology is not sufficient to meet this goal. Inflammation is a significant component of several chronic diseases involving the cardiovascular, metabolic, respiratory, gastro-intestinal, musculoskeletal and nervous systems. Positron emission tomography (PET) with targeted probes enables evaluation of the molecular and cellular pathophysiological mechanisms of diseases (i.e. molecular imaging).
However, more specific markers than the currently used glucose analogue, fluorodeoxyglucose 18F-FDG, are needed to assess inflammation and its consequences in different pathologies. Aims of the present study are pre-clinical evaluation of new targeted probes and techniques for molecular imaging of inflammation. Based on previous success, our main target is vascular adhesion protein 1 (VAP-1) that mediates leukocyte traffic into inflamed site(s) and assessment of its elevated expression in inflammation states.
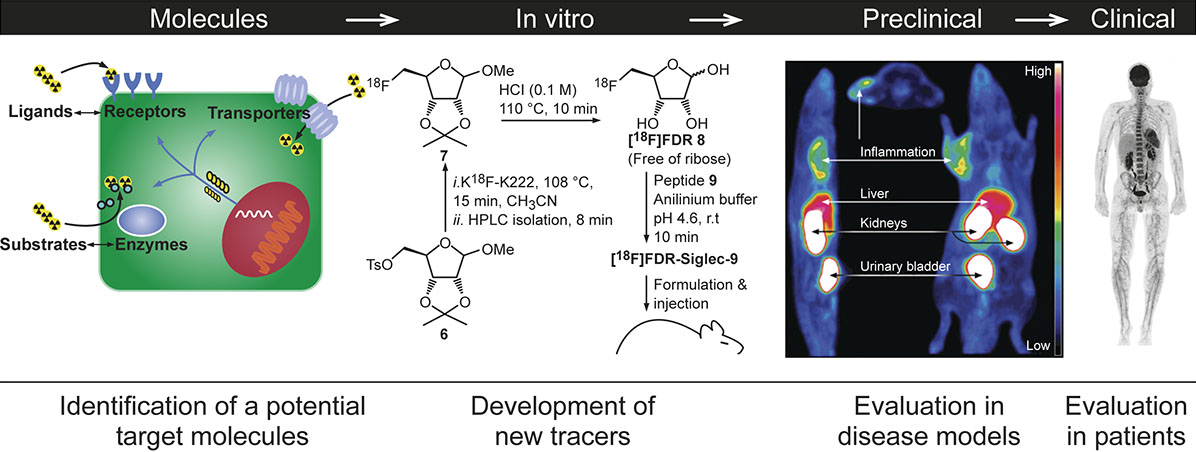
Figure 1. General development pipeline for PET-radiopharmaceuticals.
The imaging agents will be evaluated in experimental models of inflammation, in healthy subjects and in patients. VAP-1 is abundantly expressed e.g. in synovial blood vessel in rheumatoid arthritis joints. The main hypothesis is that sensitive whole-body imaging of inflammation will enable early diagnosis, help in patient selection to particular therapy and monitor therapy outcome. Noteworthy, the development of new radiopharmaceuticals requires several years’ effort and expensive proof-of-concept and acute toxicity studies. Our research is focusing on PET tracers and to lesser extent magnetic resonance (MR) and single-photon emission computed tomography (SPECT) imaging agents.
